Zeta Potential – What is it and how can it be characterised?
Zeta potential is the charge that develops at the interface between a solid surface and its liquid medium. Simply put, the surface charge of nanoparticles in solution. It is an important tool for understanding the state of the nanoparticle surface and predicting the long-term stability of a colloidal dispersion.
What? Why? And How?
Zeta potential (or Electrokinetic potential) is the potential difference between the surface of a solid particle and the surrounding liquid the particles are dispersed in. The potential difference between the particle surface and surrounding liquid media is due to the presence of charges species at the interface.
Zeta potential can be influenced by the dielectric constant of a particle and the ionic strength of the surrounding media. Therefore, when measuring the zeta potential of particles the choice of the dispersing media should be carefully considered where possible.
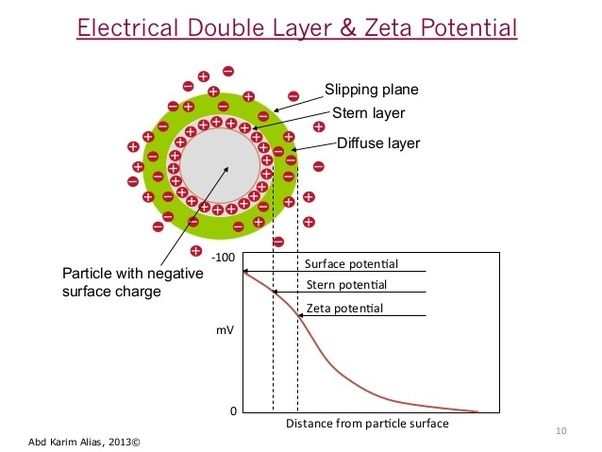
Zeta Potential Measurement
Measuring the zeta potential of particles can give valuable information on the stability of particles in solution and the potential of aggregation of particles. For biological samples zeta potential analysis can provide an insight into the cellular uptake of particles and the presence of biomarkers. A net electric charge is often present on the surface of biological particles like cells and extracellular vesicles due to their ability to absorb ions or molecules. The surface charge creates an electrical double layer around the particle which consists of a tightly bound inner layer of ions (the Stern layer) and an outer diffused layer of ions (the Gouy-Chapman layer).
Understanding the zeta potential of particles is essential in various fields of research including pharmaceuticals, cosmetics material sciences and environmental engineering.
There are several types of instruments that can be used to Measure Zeta Potential
- Electrophoretic light scattering (ELS)
- Phase analysis light scattering (PALS)
- Streaming potential instruments
- Electrokinetic analyser (EKA)
- Micro-Electrophoresis microscope
- Nanoparticle tracking analysis (NTA)


Zeta Potential Measurement Using The ZetaView
The ZetaView is a nanoparticle tracking analysis instrument, while the primary uses for the ZetaView is analysis of size, concentration and fluorescent properties of nanoparticles the zeta potential of nanoparticles can be measured by tracking the Brownian motion and the diffusion coefficient of particles in a liquid medium.
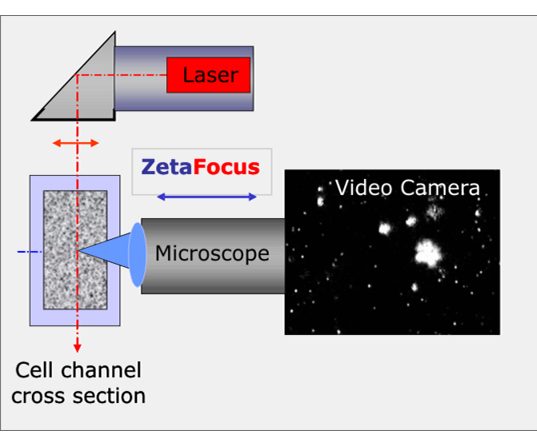
Zeta potential measurements are performed on samples loaded into a quartz glass measurement cell with electrodes located at either side of the cell. A laser and video microscope are focused throughout the measurement cell and the particles are then illuminated by the laser, the scattered light from the illuminated particles is then observed and recorded with the video microscope. Throughout the measurement cell there are 11 pre-defined positions from which measurements are taken. Two of the 11 pre-defined positions within the measurement cell are termed stationary layers, at these two layers the particles are less affected by the flow of the dispersing medium of the sample under an electric field. During the measurement an electric field is created across the measurement cell and the Brownian motion of the particles is tracked and the ZetaView analyses their trajectories, the ZetaView is then able to calculate each particles electrophoretic mobility (which can be thought of as the particles velocity in the applied electric field). Using the Helmholtz-Smoluchowski equation the zeta potential of the particles is derived from the electrophoretic mobility.
The ZetaView is able to measure the zeta potential of nanoparticles in two ways; the pulsed method and the continuous method. Both methods have advantages and limitations and the method used should take into consideration the particles to be measured and the medium they are dispersed in.
Pulsed mode measurements
Performing zeta potential measurements in pulsed mode means the electric field is created across the measurement cell intermittently in short bursts (or pulses). Using this mode the impact of electrochemical reactions and electrode polarisation can be reduced, this makes this mode particularly useful when measuring the zeta potential of particles dispersed in medium with high conductivity or ionic strength as it minimises ion migration and prevents overloading of the electrodes. Pulsed measurements can provide more accurate zeta potential values on these kind of samples as the pulsed nature of the electric field makes the result less susceptible to errors arising from polarisation effects.
Continuous mode measurements
Performing zeta potential measurements in continuous mode means the electric field created across the cell in applied in one direction continuously throughout the analysis. This method is commonly used for samples with low conductivity or when the stability and movement of particles are not significantly affected by electrode polarisation.

The ZetaView®
As the ZetaView can perform measurements using both modes this means diverse sample types can be measured using one instrument. Zeta potential can also be affected by temperature and pH, the ZetaView has a temperature-controlled measurement cell and is also compatible with a wide range of pH’s.
Zeta potential measurements can be taken at all 11 pre-defined positions or just the two stationary layer of the measurement cell. While measuring at the stationary layers in the measurement cell gives the results that are least affected by the charge of the liquid medium, taking measurements at all 11 pre-defined positions gives more statistical data (which the ZetaView corrects for the effects of the medium flow) and also provides a quality check of the measurement cell as the results should form an even parabola when plotted.
For more information on the ZetaView and how measuring zeta potential could provided valuable insights into your research contact mel.disher@analytik.co.uk


