Zika virus detectable with ASD’s LabSpec 4
Over recent years numerous outbreaks of Arboviruses, such as the Zika virus (ZIKV), have highlighted the need for proactive mosquito surveillance and rapid, cost-effective pathogen detection tests.
For the first time, research, using ASD’s LabSpec 4 shows that near-infrared spectroscopy (NIRS) is a rapid, reagent-free, and cost-effective tool that can be used to non-invasively detect ZIKV in heads and thoraces of intact Aedes aegypti mosquitoes with prediction accuracies of 94.2 to 99.3% relative to quantitative reverse transcription polymerase chain reaction (RT-qPCR).
The team estimated in this study that NIRS is 18 times faster and 110 times cheaper than RT-qPCR.

Data gathered by ASD’s LabSpec 4:
Figure 1: NIRS differentiation of ZIKV-infected and uninfected A. aegypti mosquitoes using leave-one-out cross-validation analysis with ASD’s LabSpec 4:
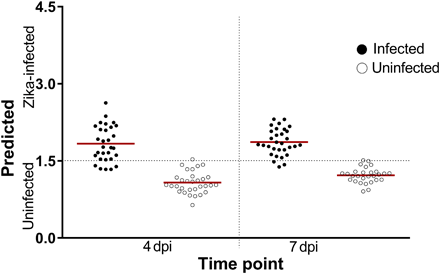
Shown are the predictions of the leave-one-out cross-validation analysis carried out on a subset of mosquitoes in cohort 1 (n = 120) at 4 and 7 dpi. Each circle represents an individual mosquito; infection status confirmed by RT-qPCR is indicated by solid (infected) or empty (uninfected) circles. Red lines indicate the mean prediction value for each group. The vertical axis indicates infection status as predicted by NIRS, with the dotted line indicating the classification cutoff point. Infected mosquitoes shown below the dotted line and uninfected mosquitoes shown above the dotted line were falsely predicted.
Figure 2: NIRS predictions of ZIKV infection in heads/thoraces and abdomens of A. aegypti mosquitoes on a blind data set:
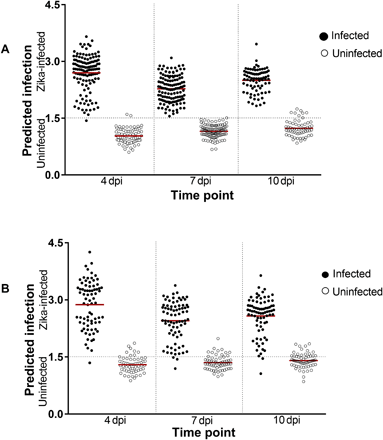
(A) Predicted infections at 4, 7, and 10 dpi in heads/thoraces for the subset of cohort 1 (n = 155) that were excluded from the model and all cohort 2 samples (n = 412). (B) Predictions for abdomens from cohort 2 samples (n = 412). Each circle represents an individual mosquito; infection status confirmed by RT-qPCR is indicated by solid (infected) or empty (uninfected) circles. Red lines indicate the mean prediction value for each group. The vertical axis indicates infection status as predicted by NIRS, with the dotted line indicating the classification cutoff point. Infected mosquitoes shown below the dotted line and uninfected mosquitoes shown above the dotted line were falsely predicted.
Figure 3: Average NIR spectra in the 350- to 2500-nm region from heads/thoraces of ZIKV-infected (red) and uninfected (blue) A. aegypti mosquitoes:
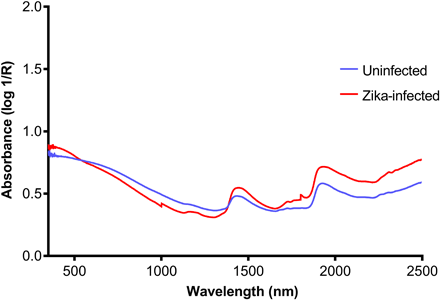
Figure 4: Regression coefficients using eight factors in the PLS model based on the NIR spectra in the 700- to 2350-nm region for differentiating ZIKV-infected from uninfected A. aegypti:
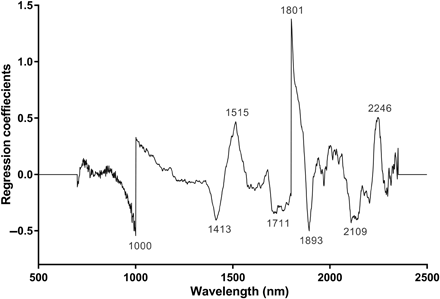
Figure 5: ZIKV load in A. aegypti cohorts at 4, 7, and 10 dpi with a Brazilian ZIKV isolate (BRPE243/2015) provided at a titer of 1.9 × 106 PFU/ml:
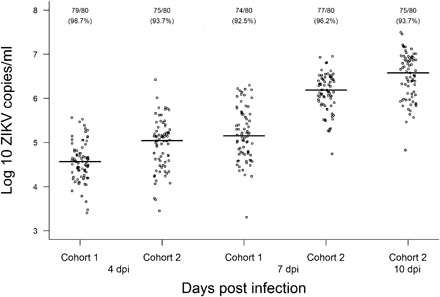
Viral copies were determined by RT-qPCR in individual mosquito homogenates with a standard curve of a seven-point dilution series (102 to 108 copies/ml) of in vitro transcribed ZIKV RNA. The number of infected mosquitoes followed by the infection rate (in parentheses) is shown for each condition.
Results:
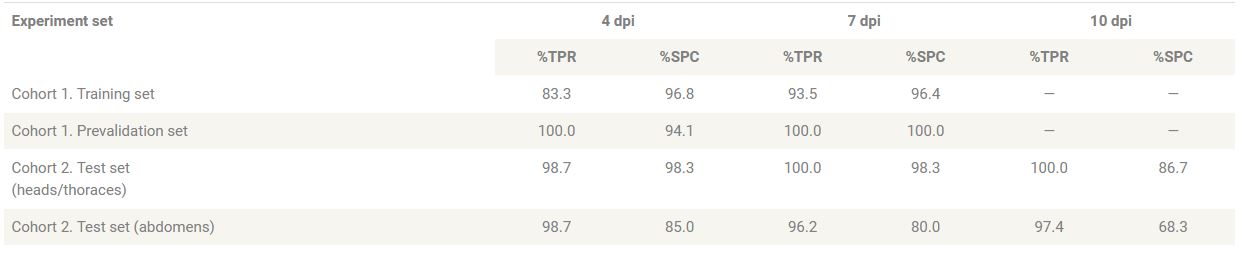
To understand whether NIRS can distinguish ZIKV-infected from uninfected A. aegypti mosquitoes, we reared adult females (cohort 1, n = 275) to 5 to 6 days of age. We fed half with ZIKV-infected blood and the other half with uninfected blood as a control group. Using ASD’s LabSpec 4 near-infrared (NIR) spectrometer, researchers collected spectra from the head/thorax region of mosquitoes at 4 and 7 days post infection (dpi) and then confirmed the ZIKV loads by RT-qPCR. They found that analysis of NIR spectra by cross-validation and partial least squares (PLS) regression distinguished ZIKV-infected from uninfected mosquitoes with 92.5% accuracy (n = 120). When they applied the ZIKV training model to a subset of samples from cohort 1 that were excluded from the training model (n = 155), they found an overall predictive accuracy of 99.3% (Figure 1). These results suggest that a randomly selected mosquito from cohort 1 could be predicted as infected with ZIKV or uninfected more than 9 out of 10 times based on the spectra of the other mosquitoes in this cohort. The sensitivity appeared to improve with increasing incubation period (Table 1).
Researchers:
- ill N. Fernandes1,*,
- Lílha M. B. dos Santos2,*,
- Thaís Chouin-Carneiro2,
- Márcio G. Pavan2,
- Gabriela A. Garcia2,
- Mariana R. David2,
- John C. Beier3,
- Floyd E. Dowell4,
- Rafael Maciel-de-Freitas2,5 and
- Maggy T. Sikulu-Lord1,†
- 1Queensland Alliance for Agriculture and Food Innovation, University of Queensland, St. Lucia, Queensland 4072, Australia.
- 2Instituto Oswaldo Cruz, Laboratório de Mosquitos Transmissores de Hematozoários, Rio de Janeiro, Rio de Janeiro 21040-360, Brazil.
- 3Department of Public Health Sciences, University of Miami Miller School of Medicine, Miami, FL 33136, USA.
- 4U.S. Department of Agriculture, Agricultural Research Service, Center for Grain and Animal Health Research, 1515 College Avenue, Manhattan, KS 66502, USA.
- 4Department of Public Health Sciences, University of Miami Miller School of Medicine, Miami, FL 33136, USA.
- 5Instituto Nacional de Ciência e Tecnologia em Entomologia Molecular, Universidade Federal do Rio de Janeiro, Rio de Janeiro 21941-901, Brazil.
This is an open-access article distributed under the terms of the Creative Commons Attribution-NonCommercial license


